A New Technique to Concentrate and Manipulate of Swimming Bacteria
4 May, 2007 12:45 pm
A surprising technique to concentrate, manipulate and separate a wide class of swimming bacteria has been identified through a collaboration between researchers at the U.S. Department of Energy's Argonne National Laboratory, Illinois Institute of technology, University of Arizona at Tucson and Cambridge University, U.K. This device could have enormous applications in biotechnology and biomedical engineering, including use in miniaturized medical diagnostic kits and bioanalysis.
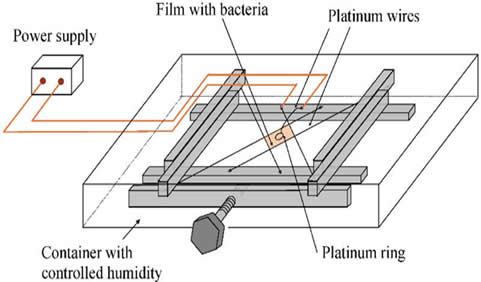
The technique is based on the transmission of tiny electric current through a very thin film sample cell containing a colony of bacteria. The current produces electrolysis that changes the local pH level in the vicinity of the electrodes. The bacteria, uncomfortable with the changes in pH, swim away from the electrodes and ultimately congregate in the middle of the experimental cell. Concentrated bacteria form self-organized swirls and jets resembling vortices in vigorously stirred fluid.
The method, which is suitable for flagellated bacteria such as E. coli, Bacillus subtilis, among many others, relies on the ability of bacteria to swim toward areas of optimal pH level. The bacteria live in an environment of a specific pH level, so that an increase or decrease of pH stimulates the bacteria to avoid areas of non-comfortable pH and swim in the direction of pH gradient. The researchers used an electric current to create a controlled deviation of the pH levels from the bulk values. Since only living bacteria respond to the pH stimulation, using this method can separate living and dead cells or bacteria with different motility. Bacteria of the length of 5 micron and swimming with the speed about 20 microns per second can be concentrated in a spot with the typical size less than 100 micron.
Figure : Layer of concentrated b.subtilis (thickness ~30micron)


Figure : Initially uniform distribution of bacteria (left), concentrated bacteria (right). Diameter of ring is 1.5mm
The device, capable to change the thickness of a film with bacteria from 1mm to 1 micron (with accuracy of 5 percent) and control the position of electrodes, is intended to separate and concentrate small quantities of living and dead microorganisms in confined spaces. It can be used for the purposes of express bioanalysis, diagnostic and identification of small bacterial samples, and separation sicken/live cells. A patent for the device is currently pending.
Unlike in traditional method for separation of particles (electrophoresis) in our experiment the applied voltage is much lower (in the range 2-3 volts). Bacteria can be separated and recognized without killing them. Moreover, the bacteria feel the concentration of ions but not the electric field. It means that they can be concentrated inside a ring due to diffusion of ions in the area where the electric field is zero.
Some bacteria (like b. subtilis) are very sensitive to a tiny change in pH level and using them as a biological pH indicator instead of standard halochromic indicator is a perfect way to find the change of acidity in a cases if we can not add any chemicals and need a great sensitivity and space resolution at the same time. These bacteria are like a bunch of sprinters which are running in different directions and measure the acidity in every corner.
Using this method, our research succeeded in dramatically increasing the concentration of microorganisms in tiny fluid drops and films. Unlike traditional centrifuging techniques, the new approach allows selective concentration of healthy cells.
In addition to the development of the device used in the experimentation, research findings uncovered the explanation for the long-standing fundamental questions on the properties of collective and organized motion in the systems of interacting self-moving objects. Besides swimming bacteria, other examples include bird flocks, fish schools, motor proteins in living cell, and even swarms of communicating nano-robots.
We have presented experimental studies of collective bacterial swimming in thin fluid films where the dynamics are essentially two-dimensional and the concentration can be adjusted continuously, explained Igor Aronson, physicists at Materials Science Division, Argonne National Laboratory. Our results provide strong evidence for the pure hydrodynamic origin of collective swimming, rather than chemotactic mechanisms of pattern formation when microorganisms just follow gradients of a certain chemical, such as nutrient, oxygen, or other.
References:
A. Sokolov, et al , Concentration Dependence of the Collective Dynamics of Swimming Bacteria, Physisal Review Letters , 98 , 158102 (2007)
I. S. Aranson, et al , Model for dynamical coherence in thin films of self-propelled microorganisms, Physical Review E , 75, 040901 (2007)






This is an interesting and well-written short article. This is a novel idea with many potential applications. However, although the reference gives relevant background theory, there is no reference to the device itself, and I cannot see the images in the article, so it is not possible for me to assess the experiments satisfactorily. A suitable reference or diagram is needed.
I accept the article in it's current form. It is a leap to selectively separate and concentrate healthy microorganisms from dead ones.